Tears do more than keep your eyes moist – they’re packed with proteins and molecules that reveal your eye health. Inflammatory markers, like IL-6, TNF-α, and MMP-9, play a key role in diagnosing and managing conditions such as Dry Eye Disease (DED), which affects millions in the U.S. These markers provide a clearer picture of cellular changes that traditional eye tests often miss.
Key takeaways:
- Markers like IL-6 and MMP-9 indicate inflammation and tear film instability.
- Testing methods include capillary tubes and Schirmer strips, each with pros and cons.
- Elevated marker levels help guide treatments, such as anti-inflammatory medications or preservative-free drops.
- Regular monitoring of these markers ensures precise adjustments to treatment plans.
Dry Eye Diagnosis | What is InflammaDry Dry Eye Lab Test? | Use InflammaDry for Dry Eye Diagnosis
sbb-itb-c87b093
What Are Inflammatory Markers in Tear Film?
Common Inflammatory Markers in Tear Film and Their Effects on Eye Health
Inflammatory markers are proteins and molecules that the body releases in response to stress or damage, signaling cellular imbalances. In the tear film, these markers include proinflammatory cytokines like IL-1, IL-6, and TNF-α, chemokines such as IL-8, and adhesion molecules like ICAM-1 and HLA-DR. When the eye experiences drying stress, the levels of these biomarkers increase, reflecting cellular changes that might not show up in routine tests.
In June 2020, researchers at the Second Affiliated Hospital, Medical College of Zhejiang University, conducted a study analyzing tear samples from 30 patients with meibomian gland dysfunction-related evaporative dry eye. Using multiplex bead analysis, they discovered that markers like ICAM-1, IFN-γ, IL-8, IL-6, TNF-α, and IL-12p70 were present in all patients. This provided clear, measurable evidence of damage to the ocular surface. Let’s take a closer look at how each marker contributes to eye health.
Common Inflammatory Markers
Each inflammatory marker has a specific role in the eye’s response to stress:
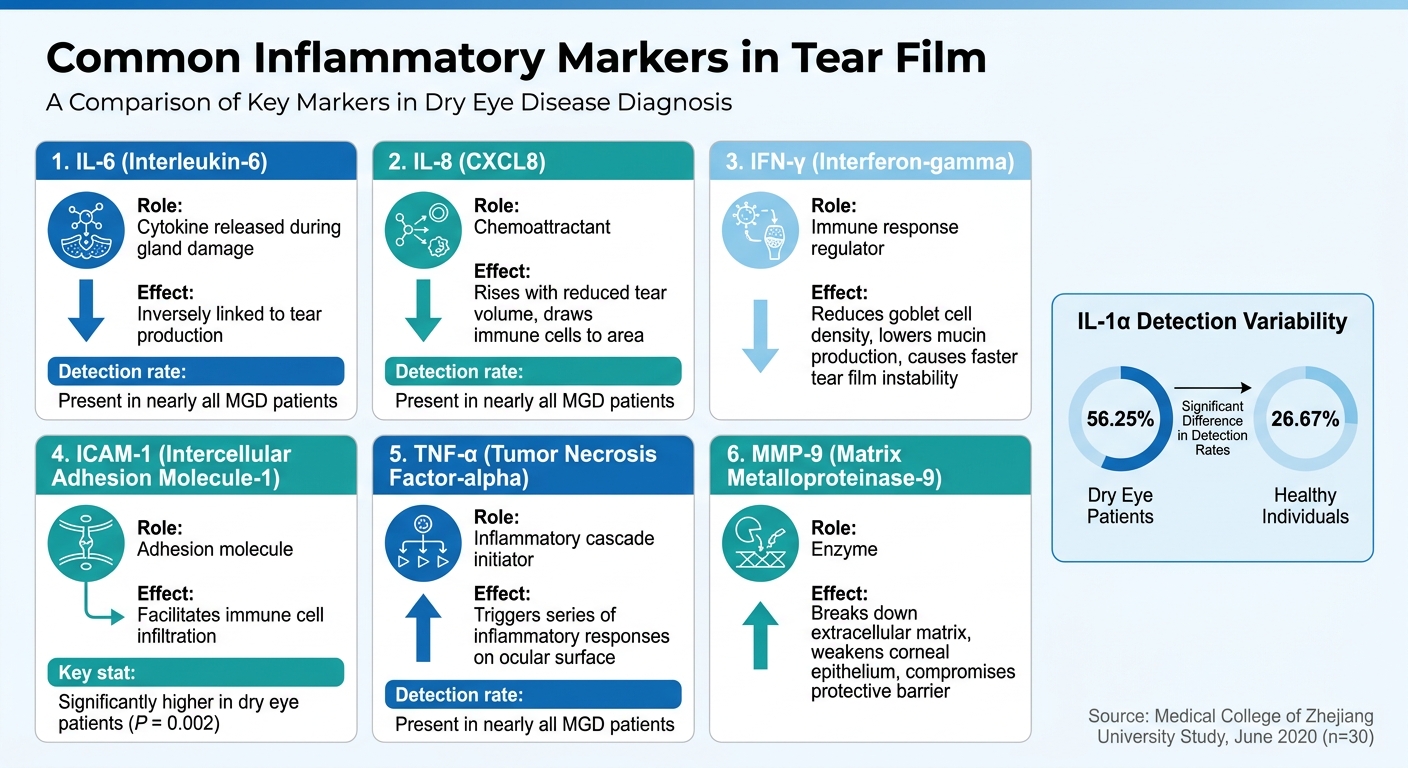
- IL-6: This cytokine increases when the glands are damaged and is inversely linked to tear production.
- IL-8 (CXCL8): A chemoattractant that rises with reduced tear volume, drawing immune cells to the area.
- IFN-γ: Reduces goblet cell density, leading to lower mucin production and faster tear film instability.
- ICAM-1: Facilitates immune cell infiltration and is strongly associated with ocular inflammation. In the study, ICAM-1 levels were significantly higher in dry eye patients compared to healthy individuals (P = 0.002).
- TNF-α: This cytokine initiates the inflammatory cascade. The study noted:
"The high expression of TNF-α initiates a series of inflammatory responses on the ocular surface, which may lead to the occurrence and development of DED".
- MMP-9: An enzyme that breaks down the extracellular matrix and weakens the corneal epithelium, compromising the eye’s protective barrier and contributing to tear film instability.
Interestingly, not all markers appear consistently. For example, IL-6, IL-8, and TNF-α were present in nearly all patients with meibomian gland dysfunction, but IL-1α was detected in only 56.25% of dry eye patients compared to 26.67% of healthy individuals.
How Inflammatory Markers Affect Tear Film
Inflammatory markers don’t just signal problems – they actively contribute to tear film instability. For instance, desiccating stress triggers the MAPK (mitogen-activated protein kinase) pathway, which leads to the production of IL-1β and TNF-α in the ocular surface epithelium. This sets off a damaging cycle: inflammation causes tissue damage, which destabilizes the tear film even further.
Take IFN-γ as an example – it reduces goblet cell density, which decreases mucin production and speeds up tear film breakup. Meanwhile, MMP-9 breaks down structural proteins that are critical for maintaining the corneal epithelium’s barrier. The combined result is a tear film that’s less stable, less protective, and less effective at supporting overall eye health. Measuring these markers not only helps identify cellular disturbances but also provides a roadmap for more precise treatments.
How Are Inflammatory Markers Tested in Tear Film?
Testing for inflammatory markers in tear film starts with the careful collection of tear samples. The two main methods used for this purpose are capillary tubes and Schirmer strips, each suited to different clinical needs and types of analysis.
Tear Sample Collection Methods
Capillary tubes are small plastic or glass tubes, typically holding about 10 µl of fluid. They are placed at the lower conjunctival fornix (the area between the lower eyelid and the eye), where tears are drawn in through capillary action. The collected tears are later retrieved using centrifugation. This method minimizes discomfort since there’s no direct contact with the cornea, but it requires precision from the clinician and takes more time.
Schirmer strips, on the other hand, are filter paper strips placed under the eyelid for five minutes. These strips absorb tears while also measuring tear volume. However, an additional elution step with a solvent like 0.9% NaCl is needed to extract proteins for analysis. While Schirmer strips are simpler to use in routine settings, they often cause discomfort and can trigger reflex tearing, which dilutes the concentration of inflammatory markers. Reflex tearing is a notable drawback, with Schirmer strips exceeding basal tear flow rates in 50% of cases, compared to 30% with capillary tubes.
A common challenge with both methods is the risk of contamination. Basal tears, which are continuously produced at a rate of 0.5–2.2 µl/min, provide the most accurate reflection of inflammation. However, irritation from the collection tools can induce reflex tears, introducing serum-derived proteins that may skew the results. For example, a study comparing the two methods found median IgG concentrations of 27.6 µg/ml in capillary tube samples versus 59.8 µg/ml in Schirmer strip samples.
Despite these challenges, both methods can yield samples that, when analyzed correctly, align with clinical test outcomes.
Analysis and Diagnostic Connections
The accuracy of tear collection directly impacts the reliability of inflammatory marker measurements, which are essential for guiding diagnostic and treatment decisions. Once collected, tear samples are typically analyzed using ELISA to detect specific proteins, such as IgG, or Bradford assays to measure total protein content. Advanced techniques like isoelectric focusing can identify patterns of inflammatory proteins, while multiplex bead analysis allows for the simultaneous measurement of multiple markers.
These molecular findings are then integrated with clinical tests to create a comprehensive diagnostic framework. For example, the wetting distance from Schirmer strips is often evaluated alongside Tear Break-Up Time (TBUT) and Ocular Surface Disease Index (OSDI) scores. When elevated levels of MMP-9 are observed along with reduced TBUT and high OSDI scores, clinicians can confirm both the biochemical and functional aspects of dry eye disease. This combination of objective data and clinical observation helps eye care professionals develop targeted treatment plans based on measurable evidence rather than assumptions.
Inflammatory Markers and Their Links to Eye Conditions
Inflammatory markers play a key role in understanding ocular diseases. They not only help explain why certain symptoms appear but also provide insights into the extent of damage and guide clinical evaluations.
IL-6 and Tear Production
Interleukin-6 (IL-6) levels are notably higher in individuals with meibomian gland dysfunction (MGD)-related evaporative dry eye. This elevation is linked to reduced tear production, as evidenced by lower Schirmer I test scores, and is associated with increased meibomian gland dropout. IL-6 serves as a useful indicator of disease severity and can also track how well a patient responds to treatment. According to International Ophthalmology:
"The level of IL-6 might be used as an indicator to define the disease severity and evaluate the anti-inflammatory drug efficacy for DED".
MMP-9 and Tear Film Instability
Matrix metalloproteinase 9 (MMP-9) is another key marker found on the ocular surface in cases of dry eye disease. It reflects inflammation and damage by breaking down proteins that are essential for maintaining a stable tear film. Elevated MMP-9 levels provide clear evidence of compromised ocular surface health.
TNF-α and Ocular Surface Damage
Tumor necrosis factor-alpha (TNF-α) is a major driver of inflammation in dry eye disease. When the ocular surface becomes dry, it activates the MAPK pathway, leading to increased TNF-α production in the surface epithelium. This marker is present in all patients with MGD-related evaporative dry eye and is responsible for widespread inflammation on the ocular surface. Identifying these markers allows clinicians to create more targeted and effective treatment strategies.
How Inflammatory Markers Guide Treatment
Personalized Treatment Plans
Inflammatory markers play a key role in crafting customized therapies. By analyzing markers in tears and blood, doctors can categorize patients more precisely – distinguishing between those with localized eye surface issues and those whose dry eye is linked to systemic conditions like autoimmune diseases. For instance, elevated levels of IL-1β, IL-6, and TNF-α suggest that Cyclosporine A (Restasis) could be effective. A 2024 trial confirmed this, showing reduced marker levels and better ocular symptoms after treatment. Daniel Friederich, a Clinical Optometrist and Dry Eye Specialist, sheds light on the broader implications:
"Serum markers… capture the patient’s systemic inflammatory milieu – including comorbid autoimmune disease and low-grade inflammation possibly aggravated by chronically absorbed ophthalmic excipients."
This data also helps identify patients who are sensitive to preservatives like benzalkonium chloride (BAK), commonly found in eye drops. Since up to 80% of an eye drop drains through the nasolacrimal duct, preservatives can enter the bloodstream and exacerbate inflammation. If inflammation persists despite treatment, doctors may review the excipients in medications and switch patients to preservative-free options to minimize toxicity.
At Boulder Eye Surgeons, these advanced assessments of inflammatory markers allow for highly personalized treatment plans. This ensures care is tailored to each patient’s specific ocular and systemic conditions, paving the way for more precise monitoring of treatment outcomes.
Monitoring Treatment Results
Baseline inflammatory markers (e.g., CRP, TNF-α, CXCL10) are measured and then reassessed every 8–12 weeks to objectively evaluate treatment progress. These markers serve as an early indicator of success, often revealing underlying issues before symptoms visibly improve.
A 2024 study from He Eye Specialist Hospital in Shenyang, China, highlights this approach. Between March and June 2024, 28 dry eye patients undergoing Intense Pulsed Light (IPL) therapy were observed. Patients who completed three IPL sessions experienced an increase in LT-α levels from 0.68 ng/mL to 1.56 ng/mL after 12 weeks, approaching the healthy range. Meanwhile, those with only two sessions remained near 0.61 ng/mL. The researchers noted:
"Research indicated that IPL therapy alters tear inflammatory cytokines, with enhancements in tear inflammation occurring prior to modifications in DED signs."
These early changes in inflammatory markers often precede noticeable clinical improvements. This helps doctors fine-tune treatment plans and determine the most effective frequency for therapies.
Conclusion
Inflammatory markers have opened a new frontier in understanding and diagnosing Dry Eye Disease (DED), offering objective and standardized insights that traditional methods often overlook. As highlighted by Springer Nature:
"Biomarkers are becoming increasingly important in DED diagnosis and drug development because they potentially provide a more standardized, objective, and precise measurement of the disease or help evaluate response to therapy".
These advancements are reshaping how DED is diagnosed and treated, enabling more precise and targeted approaches.
With tools like MMP-9 detectors and tear osmolarity devices – capable of working with less than 100 nanoliters of tear fluid – point-of-care testing has become more accessible than ever. These technologies allow clinicians to differentiate between issues like lacrimal gland dysfunction and tear film instability, ensuring that patients receive the most suitable treatments from the outset.
This progress also supports the rise of personalized treatment plans. Inflammatory markers, such as HLA-DR expression and albumin-to-lactoferrin ratios (where a ratio greater than 2:1 strongly suggests Sjögren syndrome), provide a reliable way to assess disease severity and tailor therapies accordingly.
At Boulder Eye Surgeons, the integration of inflammatory marker assessments helps deliver customized care, identify disease progression early, and improve long-term outcomes for patients, ensuring optimal eye health.
FAQs
Do inflammatory tear markers always mean you have dry eye?
Inflammatory tear markers don’t always point to dry eye. These markers can show up in a range of ocular surface conditions and might not align directly with the visible signs or symptoms of dry eye. They are just one aspect of the diagnosis and should be assessed together with other factors by your eye care professional.
How accurate are tear tests if collection can trigger reflex tearing?
Tear tests can become less reliable if reflex tearing happens during the collection process. Reflex tears may dilute the inflammatory markers in the sample, which could change its composition and affect the test’s accuracy. To achieve better results, steps should be taken to reduce reflex tearing during sample collection.
How often should tear inflammatory markers be rechecked during treatment?
Tear inflammatory markers need to be monitored regularly during treatment. How often this should happen depends on your specific situation and how your condition is progressing. It’s always a good idea to stick to the schedule your eye care provider suggests.





